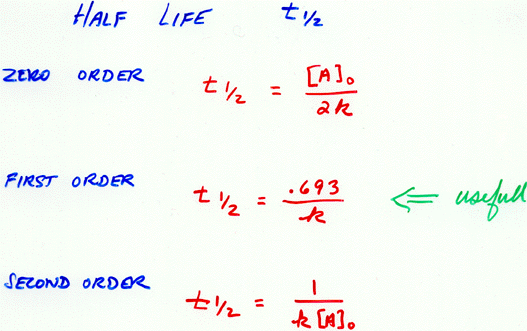
In which o is its initial concentration and k is the first-order rate constant. For a reactant A, its concentration t at time t is given by Is a common example of the law of exponential change. 
The integrated rate law for a first-order reaction A nice discussion of this property can be found here. The reason that the exponential function y=e x so efficiently describes such changes stems from the remarkable property that dy/dx = e x that is, e x is its own derivative, so the rate of change of y is identical to its value at any point. Its inverse, the law of exponential growth, describes the manner in which the money in a continuously-compounding bank account grows with time, or the population growth of a colony of reproducing organisms. Consumption of a chemical reactant or the decay of a radioactive isotope follow the exponential decay law. This general relationship, in which a quantity changes at a rate that depends on its instantaneous value, is said to follow an exponential law.Įxponential relations are widespread in science and in many other fields. The rate at which a reactant is consumed in a first-order process is proportional to its concentration at that time. First-order reactions The law of exponential change This is known as the half-life (or half-time) of the reaction. Instead of trying to identify the time required for the reaction to become completed, it is far more practical to specify the the time required for the concentration of a reactant to fall to half of its initial value. This kind of consideration is especially imporant in industrial processes in which the balances of these costs affect the profitability of the operation. The particular fraction one selects depends on the cost of the reactants in relation to the value of the products, balanced against the cost of operating the process for a longer time or the inconvenience of waiting for more product. In this case, it might make more sense to define "completed" when a reactant concentration has fallen to some arbitrary fraction of its initial value - 90%, 70%, or even only 20%. If the reaction takes place very slowly, the time it takes for every last reactant molecule to disappear may be too long for the answer to be practical. In the interest of simplicity, we will assume that this is the case in the remainder of this discussion. If the equilibrium constant is quite large, then the answer reduces to a simpler form: the reaction is completed when the concentration of a reactant falls to zero. In this case, How do we define the point at which the reaction is "completed"?Ī reaction is "completed" when it has reached equilibrium - that is, when concentrations of the reactants and products are no longer changing. How long does it take for a chemical reaction to occur under a given set of conditions? As with many "simple" questions, no meaningful answer can be given without being more precise. If you have had even a bit of calculus, here is an opportunity to put it to use! Expressing the "speed" of a reaction: the half-life  
This is easy to do, but only some courses expect you to know how to do it. This is the purpose of an integrated rate law. But for many practical purposes, it is more important to know how the concentrations of reactants ( and of products) change with time.įor example, if you are carrying out a reaction on an industrial scale, you would want to know how long it will take for, say, 95% of the reactants to be converted into products. The ordinary rate law (more precisely known as the instantaneous or differential rate law) tells us how the rate of a reaction depends on the concentrations of the reactants.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
